VDP-92 [Funding] Theranautilus
One liner: Theranautilus is developing dental stem cells with internalised magnetic bioglass nanobots for remote-controlled targeted bone growth to tackle bone loss, fracture and root canal failure.

Longevity Dealflow WG team
Shepherd: Rhys Anderson Other squad members: Tovah Wolf, Divya Cohen, Eleanor Davies
Simple Summary
Bone loss and tooth fracture are common in geriatric patients (osteoporosis/osteopenia affect around 18% of the global population). It can lead to complications if untreated. Theranautilus is a lab spin-off working to commercialize biohybrid stem cells with internalized magnetic nanobots, to allow for remote control of the stem cells inside a living body to direct them to a target location. Coating the nanobots with calcium silicate bioglass induces an osteogenic phenotype in the stem cells, allowing for targeted bone formation.
The final product will be cryopreserved biohybrid stem cells to enable easy shipment and easy storage at the hospital level along with the control device to manoeuver them.
The biohybrid nanobot technology can be applied to root canal failure. The nanobots can be remotely guided through dentine tubules to penetrate deep into the dentine to the site of bacterial infection. Magnetic fields can be applied inducing hyperthermia death. Long term, the technology can be used to target delivery of stem cells for regenerative therapy for age-related conditions e.g. skeletal bone regeneration for osteoporosis/osteopenia, hearing loss and neural regeneration in stroke patients. Teeth are ideal to test this new therapy, including the availability/accessibility of stem cells in dental pulp.
Problem
Treatment options for dental bone loss and fracture are inefficient and have high failure rates, excessive treatment times, and schedule delays. Root canal treatment failure is common, occuring in ~10% of cases (up to 5M per year in the US alone). The main cause of endodontic failure is the persistence of microorganisms that cause an intra- or extraradicular infection and become resistant to disinfection measures.
Opportunity
The Theranautilus team has been working on nanorobotics for over a decade. The team is building stem cells that can be magnetically manoeuvred to the target site due to the presence of magnetic nanostructures inside the cells. These nanostructures (CALBOTs) are coated with calcium silicate bioglass to induce an osteogenic phenotype and trigger bone growth. Stem cells are already used in many therapeutic applications and hold potential in regenerative medicine.

Theranautilus are seeking funding to translate their proof-of-concept (PoC) by generating data over a larger sample and undergoing GLP documentation for biohybrid stem cell production.
They have successfully developed the first working prototype: THERADRIVE, a miniature coil suitable for use in an adult human jaw. This is a medical device that can generate up to 60 Gauss rotating fields in any direction to drive stem cells inside the teeth.

Theradrive prototype
Cell toxicity tests The team has cell toxicity data and know the exact concentration to make the dental stem cells bioactive to trigger bone growth whilst keeping them viable for over a month. This has been validated on mouse preosteoblasts (MC3T3 E1 subclone 4 mouse calvarial pre-osteoblasts (ATCC, USA) ).
Bone growth tests After establishing cell cytotoxicity and confirming the magnetic bioglass is not cytotoxic, they established the bone growth potential of their nanomaterials.

Representative images of bone growth localized to the regions where CALBOTS are present
Workflow Dental Pulp Stem Cells (DPSCs) will be extracted from wisdom teeth and chilled in HBSS for up to 12 hours. The laboratory will perform quality control on the DPSCs followed by cryopreservation. When required for therapy, the cells will be thawed and expanded, incubated with CALBOTs, before returning to the patient.
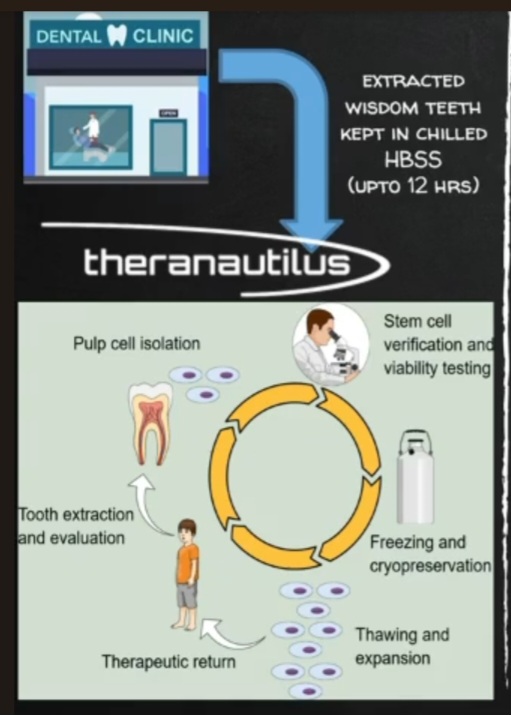
Theranautilus workflow
For root canal failure, the nanobots can be guided down the dentinal tubules, followed by application of rapidly varying magnetic fields to heat the nanobots and cause hyperthermia-induced death in bacteria. The team has optimised the procedure for root canal in a rat, including miniaturisation of root canal instruments to custom fit the rat’s oral cavity. Successful root canal procedures have been performed on the rats and a protocol has been developed for upcoming randomized controlled trials with a research hospital.
Estimated treatment cost analysis (in USD)
Refer to VDP on Discourse for references and cost analysis breakdown
Current Cost of stem cell banking: a) Initial Processing : $500 - $2,000 b) Annual Maintenance (Recurring): $99 - $264
Breakdown of estimated cost for modifying stem cells: a) Materials cost (consumables – One time cost per patient): $50 - $100 b) Service Charge (Optional; In case Theranautilus provides lab support; cost per patient): $20
Experimental plan and budget
Pre-Clinical Studies 1a: Toxicity assays of magnetic bioglass on mesenchymal human stem cells (MSCs).
To perfect the protocol of incubating MSCs with magnetic bioglass and the optimum concentration of magnetic bioglass to be used. Cell viability assays at different day points – days 1, 4, and 7. Similar experimental data has been done on mouse pre-osteoblasts, and the same protocol will be followed with necessary adjustments.
- Required Funding: $31,210
- Duration: 3 Months
Pre-Clinical Studies 2a: Efficacy of magnetic bioglass in triggering bone growth in mesenchymal human stem cells. To test if bone growth is happening with spatial control, by demonstrating the magnetic manoeuvrability of stem cells to target bone fracture and the possibility of patterned bone growth.
- Required Funding: $71,465
- Duration: 6 Months
Pre-Clinical Studies 1b: GLP certification for magnetic bioglass fabrication. This is the next step before applying for clinical trials. The magnetic bioglass synthesized by Theranautilus’ protocol will undergo GLP certification before human trials, where the team plans to collaborate closely with a GLP certified lab to provide this certification.
- Required Funding: $38,150
- Duration: 6 Months
Pre-Clinical Studies 2b: Device design to deliver mesenchymal stem cells in the human teeth. The device is protected by the patent application (https://patents.google.com/patent/US20220226073A1/en). The team will look at making minor modifications to their current control algorithm and machine to work with CBCT or x-ray data to drive magnetic maneuverable stem cells to the fracture site. An image processing component may be added to the device to make it more efficient.
- Required Funding: $43,000
- Duration: 6 months
Total budget: $183,825
VitaDAO Funding Terms
$183,000 (USD) via Sponsored Development Agreement for 15.25% of the Net Company Receipts (valued at $1.2M based on equity raise), encompassing the following for CALBOTs therapies: bone loss, tooth fracture, root canal failure. This would be brought on-chain as an IP-NFT.
IP Roadmap
Theranautilus owns the IP to manufacture the devices to control their nanostructures in human teeth. They filed the composition of matter patent in 2022 and will file a PCT for protection in US/EU. The patent protecting the method of creating biohybrid stem cells will be filed once the bone growth data and toxicity experiments are repeated.

Team
Dr. Debayan Dasgupta - Co-founder, Director Dr. Shanmukh Srinivas - Co-founder, Director Prof. Ambarish Ghosh - Co-founder, Director Full profiles on Discourse
Strengths
- The team are pioneers in implementing magnetic manoeuvrability of nanostructures in biological systems
- Current medical procedures cannot reach the required depths to kill bacteria. Team has demonstrated PoC drug delivery using nanobots in dentine tissue of human teeth
- Successfully demonstrated drug delivery inside live Wistar rats using nanorobotics technology
- Quick route to market
- Teeth are ideal for testing new cell-based treatments. Dental pulp is one of the few areas where stem cells are still found in human adults. Could provide PoC to expand into other regenerative therapies
Risks
- Risk associated with novel technology
- Nanobots can be magnetically guided down dentinal tubules but no evidence for stem cell hybrids yet
- Adoption of the technology to replace current therapies may be difficult
Senior Review Digest - Quantitative
Below are the average scores out of 5 per category from 4 reviewers
Average Scores
- Novelty & impact: 4
- Feasibility & data: 2.75
- Relevance: 3.5
- Science team: 3.5
- Market advantage: 3
- IP Potential: 4.5
- Conviction: 2.75
Read the Discourse VDP for the full review and references.
Off-Chain Vote
Timeline
Jan 28, 2024Proposal created
Jan 29, 2024Proposal vote started
Feb 05, 2024Proposal vote ended
Nov 18, 2024Proposal updated