VRNAP-4 Token Mint Proposal for $1M Research Funding
VRNAP-4 Token Mint Proposal for $1M Research Funding
We propose minting additional VitaRNA tokens equivalent to $1,000,000 to advance our program toward IND-enabling studies.
Token Mint Details
Amount: $1,000,000 worth of VitaRNA tokens. The exact amount of tokens will be determined based on the final auction price which will be determined by the time the sale is concluded.
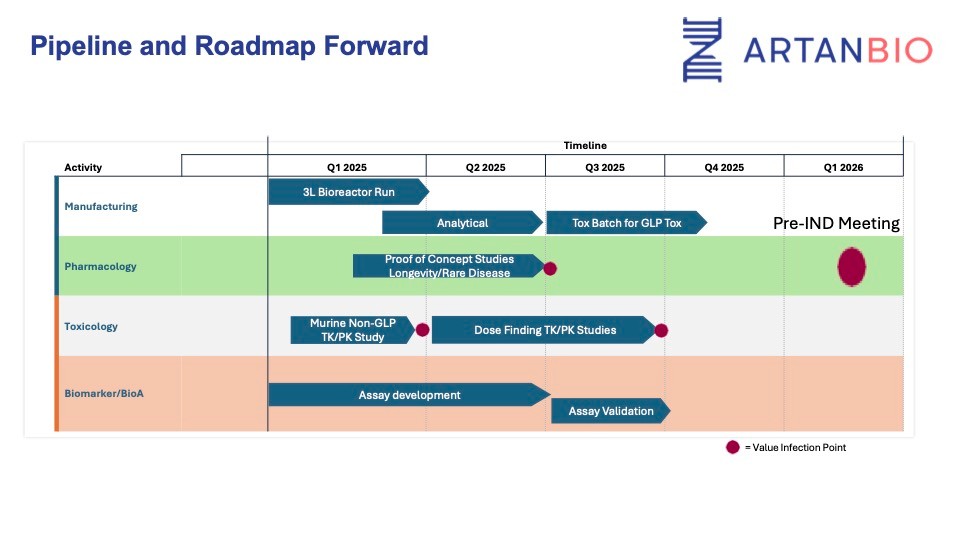
Purpose: Fund IND-enabling studies and related development activities
Inflation: App. 3.5% at current prices
Use of funds:
-
Lonza manufacturing to support GLP-tox studies (required to do a clinical trial)
-
Bioanalytical assay development (needed to support GLP tox and human clinical trials)
-
Biomarker validation to support a longevity trial
-
Additional safety studies
-
Biomarker animal studies (longevity)
-
Further patent prosecution
-
FDA Consultant
-
Clinical/GLP AAV Consultant
-
General and administrative (G&A)
Target deliverables:
-
Drug product suitable for GLP-tox studies
Lay explanation: FDA requires drug candidates to be manufactured and characterized in a specific way. Lonza would make material in way that acceptable to FDA for GLP-tox studies, which are the animal studies required by FDA to approve testing in humans
-
Biomarker assay that could support a human clinical trial
Lay explanation: A clinical trial that looks at lifespan as an endpoint is not currently feasible (takes too long). Therefore we will work with partners (like Tempus or Natera) to develop a set of biomarkers based on arginine mutations and an assay to demonstrate Artan-102 has an aging impact. This is a lofty goal, but important. This is most critical, but hardest thing for us to do as it relates to human clinical trials.
-
A data package suitable for a pre-IND meeting with the FDA
Lay explanation: For gene therapy products, it’s not uncommon to have a pre-IND meeting with FDA to align on the study designs needed by FDA to support a human clinical trial.
-
Key milestone for all this work is to have a pre-IND meeting with FDA.
This would be an amazing milestone for the DeSci community! A fully DeSci grown project that makes it the FDA for consideration!
Target Timeline for the $1M raise
Off-Chain Vote
Timeline
Dec 06, 2024Proposal created
Dec 06, 2024Proposal vote started
Dec 08, 2024Proposal vote ended
Mar 07, 2026Proposal updated